How EU’s PPWR Impacts US Medical & Biotech Exports
For US manufacturers of medical devices, diagnostics, and biotechnology products, the European Union (EU) represents a critical market. However, access to that market now requires navigating the complex new reality of the Packaging and Packaging Waste Regulation (PPWR), which aims to make packaging in the EU sustainable.
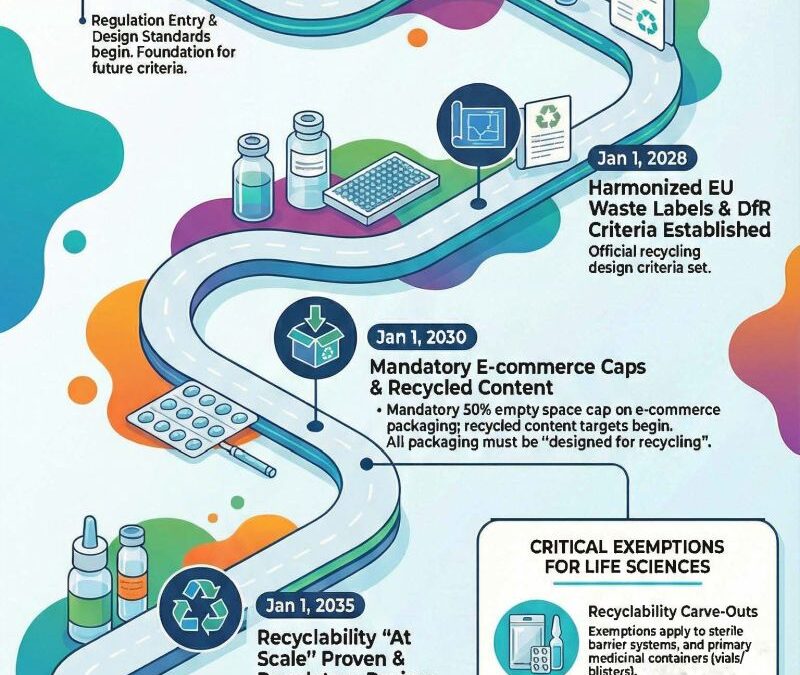
Key dates about PPWR:
- February 20, 2025: Regulation officially entered into force.
- January 1, 2028: Criteria for Design for Recycling (DfR) to be established
- January 1, 2030: Packaging must be “designed for recycling,” and empty space in e-commerce boxes must be capped at 50%.
- January 1, 2035: Recyclability must be proven “at scale” (meaning the infrastructure to actually recycle the specific material must exist in the EU).
- 2050: Target climate neutrality
For exports to Europe from the US, here are the major hurdles to clear:
|
Requirement |
What it means |
|
PFAS (“forever chemicals”) Ban |
Reduce PFAS to required trace levels in packaging by 12AUG2026. |
|
Recyclability |
By 2030, all packaging must be recyclable with exceptions mentioned below. Shipping systems and outer packaging should be reviewed against PPWR. EU will be working towards setting up infrastructure for recycling by 2035. By 2038, it must meet high-performance “Grade A or B” standards to stay on the market. |
|
Recycled Content |
Plastic packaging must contain a minimum percentage of post-consumer recycled content (e.g., 30% for most plastics by 2030). |
|
Labeling |
Companies will need to use harmonized EU labels that tell consumers exactly how to sort the waste (expected around 2028). |
|
Authorized Rep |
Non-EU companies selling directly to consumers must appoint an Authorized Representative in the EU to handle compliance. For more information on importers and distributor requirements, refer to Articles 18 and 19 respectively. |
Some exceptions for the recyclability requirements apply for the following:
- Primary packaging of human medicinal products (Article 1, point (23), of Directive 2001/83/EC) (e.g., vials for vaccines, blisters for tablets, etc.) with specific requirements to preserve the quality of the medicinal product
- Medical device sterile barrier system and primary packaging (Regulation (EU) 2017/745)
- Contact-sensistive packaging of in vitro diagnostic medical devices (Regulation (EU) 2017/746)
These exceptions will be reviewed by 2035.
Conclusion
Although many of the deadlines are years away, reviewing the packaging currently in use should begin as soon as possible. Implementation of packaging changes may take a few years because of all the qualifications and associated documentation that need to be completed.
What can Lean Biologix (LBX) do to help?
The LBX team has extensive experience with selecting and qualifying packaging and shipping systems for a variety of biotechnology products that require temperature control. Temperature requirements include ultra cold (< -60℃, < -65℃, < -130℃, etc.), cold (-20℃), refrigerated (2-8℃), and ambient temperatures. LBX can develop, evaluate, and execute packaging and shipping qualifications for a customer’s review while being compliant with the current industry and regulatory standards for the desired market, such as EU. For more information on shipping qualification, see the Design Qualification (DQ), Operational Qualification (OQ), and Performance Qualification (PQ) tabs under Supply Chain Testing Services on the LBX website.