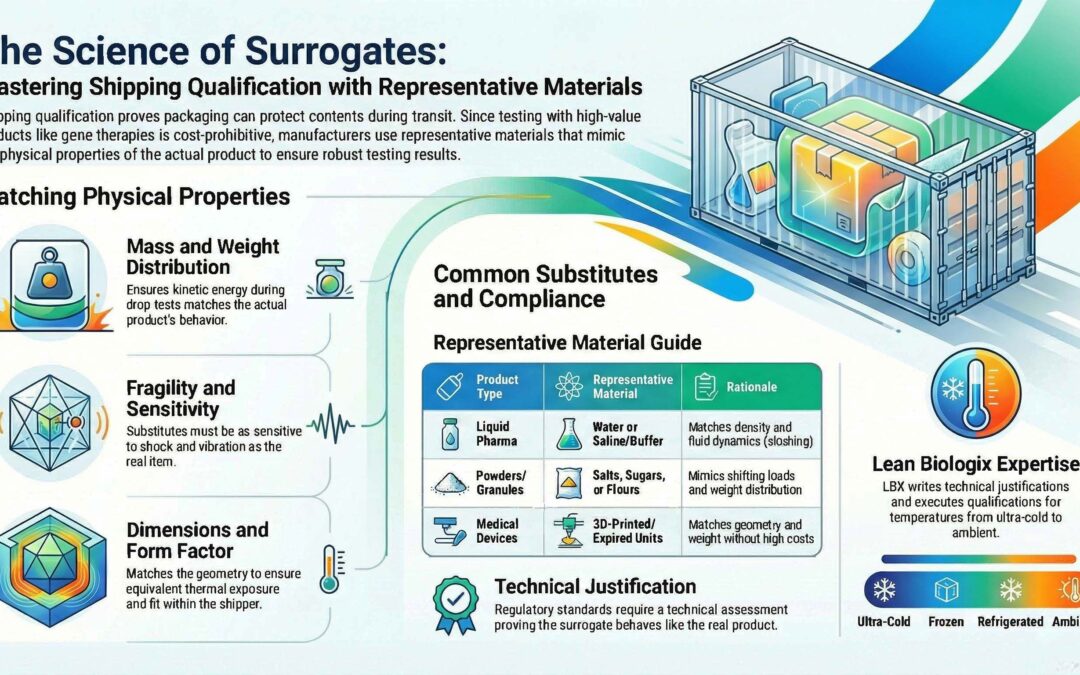
Selection of Representative Materials for Shipping Qualification
Shipping qualification is the rigorous process of proving that a packaging system can protect its contents through the hazards of the supply chain. A critical question arises during this process: Do we need to test with the actual product, or can we use representative material?
Using high-value products (like gene therapy products) for destructive testing is often cost-prohibitive. Instead, engineers use representative materials—surrogates that mimic the physical properties of the real product, when possible.
Defining “Representativeness”
To select an appropriate substitute, you must identify the attributes of the product that affect packaging performance. The goal is to match the surrogate to the product’s physical behavior, not its chemical function.
Key Physical Properties to Match:
- Mass and Weight Distribution: Ensures the kinetic energy during a drop test is identical.
- Fragility: The substitute should ideally be as sensitive (or more sensitive) to shock and vibration as the actual item.
- Dimensions/Form Factor: Substitute should fit equivalently into the shipper to withstand equivalent shocks, vibrations, and thermal exposures within the shipper.
Common Representative Materials
The table below shows a couple common “dummy” materials used for different product types in shipping qualifications:
|
Product Type |
Representative Material |
Rationale |
|
Liquid Pharmaceuticals |
Water or Saline/Buffer Solutions |
Matches density and fluid dynamics (sloshing). |
|
Powders/Granules |
Salts, Sugars, or Flours |
Mimics shifting loads and weight distribution. |
|
Medical Devices |
3D-Printed Models or Expired Units |
Matches the geometry and weight without the cost of sterile components. |
Technical and Regulatory Justification
If a representative material is chosen, a technical assessment is recommended for justification for the substitute material that behaves similarly to the real product under stress.
For example, if testing a vaccine, a Glycol-Water mixture may be used to mimic the viscosity and thermal properties of the vaccine, ensuring that the thermal profile during a 72-hour cold-chain qualification is accurate.
Conclusion
Selecting the right representative material is a balance of risk management and cost-efficiency. By focusing on mass, geometry, and fragility, companies can execute robust shipping qualifications that provide high confidence in product safety without sacrificing expensive inventory.
What can Lean Biologix (LBX) do to help?
The LBX team has extensive experience with selecting and qualifying shippers for maintaining ultra cold (< -60℃, < -65℃, < -130℃, etc.), cold (-20℃), refrigerated (2-8℃), and ambient temperatures. LBX can develop, evaluate, and execute shipping qualifications for a customer’s review while being compliant with the current industry and regulatory standards. LBX has helped write technical justifications for acceptable representative materials for shipping qualification use. For more information on shipping qualification, see the Design Qualification (DQ), Operational Qualification (OQ), and Performance Qualification (PQ) tabs under Supply Chain Testing Services on the LBX website.