
by Lean Biologix | Aug 22, 2025 | Supply Chain Testing
ASTM D4169 vs. ISTA Distribution Tests Two (2) prominent organizations lead the way in developing distribution standards: the American Society for Testing and Materials (ASTM) and the International Safe Transit Association (ISTA). This article will compare ASTM D4169,...

by Lean Biologix | Jun 19, 2024 | Computer System Validation
The GAMP5 second edition improved upon the original edition from 2008 and was released in July 2022. The updated guide keeps the same structure but updates its content to highlight the growing area of cloud services. It also addresses the increasing use of testing...

by Lean Biologix | Jan 12, 2024 | IT Project Management
Continuous Computer Software Assurance (CCSA) The modern computer software assurance model champions a computer system validation program with effective risk assessment processes. Life science companies have streamlined processes and are focused on meaningful testing...

by Lean Biologix | Nov 19, 2023 | Supply Chain Testing
Why choose a pre-qualified shipping system? At some point when designing a supply chain, a system for transporting the product must be developed or selected from available shipping system vendors. The most straightforward and least time intensive option is selecting a...
by Lean Biologix | Nov 19, 2023 | Supply Chain Testing
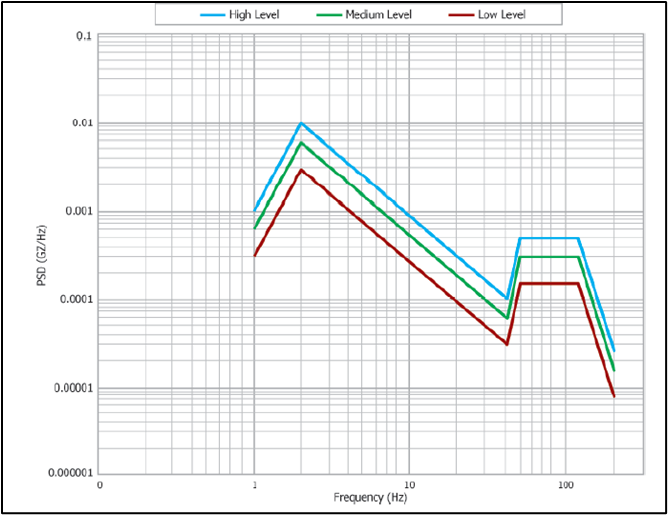
What is ASTM D4169 testing? ASTM International developed the standard ASTM D4169, “Standard Practice for Performance Testing of Shipping Containers and Systems,” to test a shipping systems’ ability to withstand typical distribution hazards that occur in a supply...






